Does badger vaccination reduce TB in cattle?
 The injectable badger BCG vaccine (delivered to badgers caught in cage traps) has been licensed for use in badgers since 2010, based on evidence that the vaccine is safe and has a protective effect in badgers (Brown et al. 2013). Captive trials have shown that vaccination reduces the development and progression of disease (Chambers et al. 2011). Results from field trials also demonstrate that vaccination reduces the spread of disease within badger populations, as vaccinated badgers are much less likely to become infected with TB (Carter et al. 2012). Crucially, vaccination is also safe, the BCG bacteria is not excreted by badgers and the vaccination process does not cause perturbation (Lesellier et al. 2006; Woodroffe et al. 2017).
The injectable badger BCG vaccine (delivered to badgers caught in cage traps) has been licensed for use in badgers since 2010, based on evidence that the vaccine is safe and has a protective effect in badgers (Brown et al. 2013). Captive trials have shown that vaccination reduces the development and progression of disease (Chambers et al. 2011). Results from field trials also demonstrate that vaccination reduces the spread of disease within badger populations, as vaccinated badgers are much less likely to become infected with TB (Carter et al. 2012). Crucially, vaccination is also safe, the BCG bacteria is not excreted by badgers and the vaccination process does not cause perturbation (Lesellier et al. 2006; Woodroffe et al. 2017).
For a summary see the badger vaccination fact sheet, and for more information on perturbation read the fact sheet on the RBCT. The reviews by Chambers et al. (2014) and Godfray et al. (2013) also provide a good summary of the science on the subject.
Logically reducing disease spread in badger populations can only be a good thing for cattle, but are there any studies which prove one way or the other that vaccination does or doesn’t ‘work’?
The short answer is no, there are no specific scientific trials or experiments that have been conducted with the explicit aim of investigating the effect of badger vaccination on TB in cattle. Such a trial would ideally need to be randomised (areas randomly selected to avoid potential bias), replicated (carried out in several locations to demonstrate how effects might vary) and with controls (matched comparison areas without vaccination), which was the case in the RBCT.
However, there are two areas in the UK where large scale badger vaccination has been conducted, these are the BVDP and IAA, which are often mentioned in discussions about badger vaccination.
The Badger Vaccine Deployment Project (BVDP) – England
This was a 100 km2 (approx.) area in Gloucestershire. Badgers vaccination was conducted from 2010 – 2015 by APHA staff, with an average of 7.6 badgers per km2 vaccinated each year (range = 5.4 to 10.0). The aims of the BVDP are outlined in the BVDP lessons learned report (APHA 2015):
“The BVDP was not set up as a scientific trial to investigate the effect of BadgerBCG deployment on TB in badgers or cattle. Rather, it aimed to provide the first use of a vaccine for bovine tuberculosis in badgers outside of research trials, to develop practical knowledge on the processes involved in vaccinating badgers and to facilitate training of lay vaccinators” – page 3.
The Intensive Action Area (IAA) – Wales
The IAA was a 288 km2 area primarily in Pembrokeshire, but with land also in Ceredigion and Carmarthenshire. The area was selected for a range of extra disease control measures due to having one of the highest rates of TB in Wales, as well as evidence of TB in the local badger population. The IAA was not a vaccination trial, a whole range of measures aimed at cattle and wildlife were applied to the IAA starting in 2010:
- stricter cattle controls and testing
- improved biosecurity measures
- testing all goats and camelids
- badger vaccination (2012 – 2015, delivered by government)
For more information see https://gov.wales/intensive-action-area-iaa
Approximately 3.9 – 4.9 badgers per km2 were vaccinated in the IAA each year. A road kill survey of badgers also recorded low levels of TB in the badger population, with the number of positive badgers declining during the project, but the sample size was too low to show a significant difference (APHA 2016).
Although neither of these areas were established as scientific studies to investigate the effects of badger vaccination, APHA has produced reports looking at changes in TB in cattle in both areas.
What do these reports tell us about the effectiveness of badger vaccination?
The BVPD report
Click here to download and read the full BVDP report
The BVDP report investigated changes in cattle TB incidence rate (the rate of new breakdowns) in the BVDP and four comparison areas. These four comparison areas were selected using a process to identify areas similar to the BVDP in their TB history and cattle herd demographics.
The analyses conducted to investigate changes in TB in the BVDP and comparison areas was descriptive and relatively simple, in that the areas were not directly compared in a single analysis (ie vaccinated vs non-vaccinated, as was used during the RBCT). Rather separate individual analyses were conducted for each area and the trends (increasing or decreasing) were then described. The severe limitations of the analyses meant that it was unlikely any effects of badger vaccination would be detected, as clearly stated in the report:
“It is important to remember that this is a descriptive analysis, and that it is unlikely that any significant changes will be observed. Any analysis of the effect of vaccination is limited by the retrospective and non-randomised selection of comparison areas. In addition, the BVDP was only conducted in one intervention area so it is anticipated that there is the potential for selection bias and that there will be insufficient power to detect the effects of vaccination on cattle incidence in the area.” (page 5)
Did TB change in the BVDP area?
The TB incidence rate in cattle significantly declined throughout the course of the BVDP, with a reduction in OTF-W (official TB free withdrawn) breakdowns of more than 50% by the end of the five year project. However, there were also significant declines in three of the four comparison areas (see figure). This could suggest that the decline in the BVDP would have happened without vaccination, but it is difficult to be sure using the analyses used.

Trends in OTF-W incidence per 100 herd years at risk over time in the central areas of the BVDP area and the comparison areas (dashed lines indicate the start of vaccination).
For example, when analyses similar to those in the BVDP report were applied to the Gloucestershire and Somerset cull areas (which are both 2-3 times larger) they found no effect of culling. Effects of culling were ONLY detected using much more sophisticated multivariable analyses (controlling for other non-cull effects) directly comparing the cull areas to TEN comparison areas (Brunton et al. 2017, read the fact sheet here). This highlights the importance of robust analyses, and the risks of coming to false conclusions if they are not conducted properly.
One other major problem is that the BVDP is a single area, and it is unclear whether this is representative of the wider landscape in relation to the role of badgers, or other factors relevant to TB (as many other factors influence TB risk ). TB was declining prior to vaccination in the BVDP and the similar matched areas experienced a drop in TB without any specific intervention. This could suggest that other non-wildlife factors were important in these areas, or changed during the trial.
The limitations of the analyses are described in detail in the report. The authors also make it clear that it would be wrong to come to firm conclusions about the general impacts of vaccination:
“The factors discussed here make assessing the impact of badger vaccination on cattle bTB incidence difficult and drawing firm conclusions from the data analysed here on the impacts of badger vaccination inappropriate.” – page 15
The IAA report
Click here to download the full IAA report
As with the BVDP report, the analyses conducted on the IAA were quite simple and descriptive in scope (ie they describe trends but make no attempt to identify the factors causing them). Trends in TB incidence (breakdown rate), prevalence (% of herds restricted) and a wide range of other TB metrics were compared for the IAA and a comparison area in west Wales.
Did TB change in the IAA?
Because of the range of different statistics calculated it is difficult to give a simple answer to this. Some metrics showed an improvement, for example the proportion of herds under restrictions in the IAA (prevalence) fell from 22-30% in the early part of the study to 14% by 2016. There was also a 35% reduction the TB incidence in the IAA, relative to a 22% reduction in the comparison area (these differences were reported by farminguk and the BVA). However the authors of the report do state that “Notable differences in indicators of TB incidence have not yet been seen” (as of 2016).

Herds under restriction because of loss of OTF status in the IAA and comparison area. Taken from report.
The IAA report makes no statement on the effects of badger vaccination, partly because vaccination was used alongside a wide range of other measures, which makes it impossible to determine which specific measures did or did not work.
Another reason it is difficult to draw conclusions is because the IAA is a quite different to the comparison area used in the analysis. This can be seen in the figure above, as the prevalence in the comparison area is much lower than the IAA. This issue is also further outlined by the authors:
“Selection of the IAA and the CA (comparison area) was not performed randomly, nor were either area replicated. There are known demographic differences between the IAA, the CA and the other areas which might influence TB trends in those regions independently of policy interventions. For example, there are a higher proportion of large dairy herds in the IAA than in other areas, and such herds are considered more likely than beef herds to see TB incidents, regardless of location. Consequently, it is not appropriate to use conventional statistical tests when making comparisons because the areas are not directly comparable.” – page 8
This is not surprising, as the IAA was not chosen to be representative of the wider welsh countryside. It was chosen due to the high levels of TB and for other differences which make controlling TB challenging, hence the ‘intensive’ approach taken. By definition the outcome of any intervention in the IAA is likely to be quite different to other areas, with different levels of TB in badgers and cattle.
Conclusions
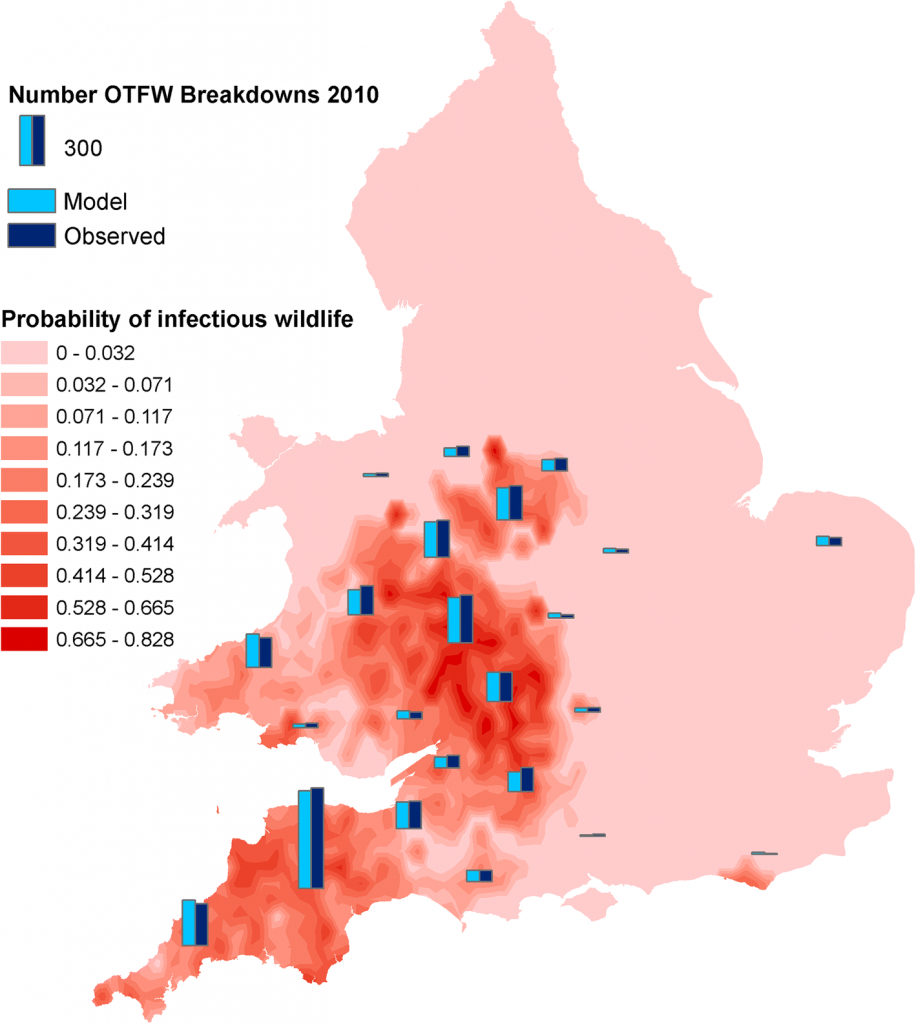
Predicted distribution of infectious environmental reservoirs (including wildlife), as estimated by modelling. Figure taken from Birch et al. 2018. Click image to enlarge.
So what can the IAA and BVDP tell us about the effect of badger vaccination on TB in cattle? Unfortunately very little. In both areas there was evidence of TB declining in the cattle population, but it is not possible to say whether vaccination contributed to this or not.
The authors of both the IAA and BVDP reports make it very clear that neither of these projects were intended as a scientific studies to prove or disprove the effects of badger vaccination. The authors also clearly outline several reasons why it would be wrong to use the descriptive analyses conducted to come to firm conclusions about vaccine effectiveness. Proving the effect of any intervention, whether its culling, vaccination, biosecurity or other cattle measures is extremely difficult, either requiring large replicated controlled trials (like the RBCT), or robust sophisticated statistical analyses. The contribution of badgers to TB in cattle likely varies hugely across the UK (Birch et al. 2018). This also means that impacts of vaccination (or any intervention) will also likely vary, and that the results from one area may not apply to other areas where the situation is very different.
Until further trials or analyses are conducted it is not possible the say definitively what effect badger vaccination will have on TB in cattle. There is however, good scientific evidence that vaccination is an effective means to reduce transmission and disease spread in badgers (as summarised here). If the aim is to have fewer infected badgers in the landscape, then vaccination is a potential tool to achieve this. Vaccination is also a viable option in parts of the country where the badger population is currently free of TB, as a way of reducing disease spread and future risk to cattle.

 @andy_1robertson
@andy_1robertson


